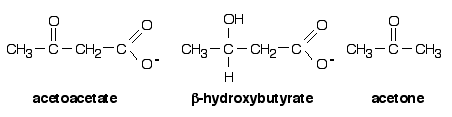
Figure 1: Chemical structures of the ketone bodies
Rapid analytical techniques for diagnosing and managing diabetes: Theory
4.1 Introduction
The aims of this practical are:
4.2 Theory and background
A detailed discussion of diabetes is beyond the scope of this short introduction. A good account can be found in the "ABC of Diabetes" by P.J. Watkins (4th edition, 1998), BMJ Publishing Group and "Diabetes: Pocket Picture Guides" by H.J. Bodansky (1989) (J.B. Lippincot).
Primary diabetes mellitus is divided into two forms: insulin-dependent diabetes (Type 1, or IDDM) and non-insulin-dependent diabetes (Type 2, or NIDDM). The former results from the destruction of pancreatic B cells in the islets of Langerhans with the consequent loss of insulin production while the latter has many possible causes leading either to lowered insulin secretion or increased resistance to the action of insulin – mostly due to a loss of insulin receptors in normally insulin-responsive tissues. Differential diagnosis is important for the proper treatment and management of the disease. In all cases, however, hyperglycaemia is present and an elevated glucose concentration can be detected in both blood and urine (glucosuria) due to the inability of the tissues to take up glucose (an insulin-dependent process). Typically, an elevated fasting blood glucose (>7 mM) or a randomly taken blood glucose > 11 mM indicates diabetes.
Measurement of blood glucose in a glucose tolerance test may be done in equivocal cases. However, measurement of urinary glucose is also a good indication of diabetes and can be combined with other urine tests.
Tests for sugars
For proper diagnosis, it is important that the method used is specific for glucose, since an elevated level of other sugars in the urine (glycosuria) may indicate quite different disorders e.g. fructose intolerance or galactosaemia.
Ketone bodies
Most of the acetyl-CoA produced by fatty acid oxidation in the liver is fully oxidised in the mitochondrial citric acid cycle (also known as TCA or Krebs' cycle) to CO2 to yield energy. Some, however, is converted to acetoacetate in a process known as ketogenesis . This acetoacetate can then be enzymically reduced to b-hydroxybutyrate or non-enzymically decarboxylated to acetone and CO2 (Fig. 1) Collectively, these three compounds are known as ketone bodies and are the water-soluble equivalent of fatty acids. Acetoacetate and b-hydroxybutyrate are released from the liver and carried in the bloodstream to peripheral tissues such as cardiac and skeletal muscle where they serve as important alternative fuels.
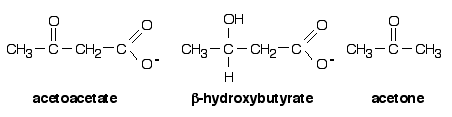
Figure 1: Chemical structures of the ketone bodies
Another symptom of uncontrolled diabetes is ketosis, where acetoacetate is produced faster than it can be metabolised. This is due to the greater than normal reliance on the breakdown of lipid stores to provide energy due to the inadequate supply of absorbed carbohydrate. Ketosis also occurs during fasting (starvation), and under these conditions ketone bodies become the brain's major fuel source. However, as ketone bodies are water soluble, significant amounts are lost in the urine and represent a loss of calories in addition to those lost as glucose. If IDDM is left undiagnosed and untreated, or if insulin is withdrawn for some reason, then diabetic ketoacidosis (DKA) can occur. Since acetoacetate and b-hydroxybutyrate are acids, elevated blood concentrations overload the buffering capacity of the blood and the kidneys, which control blood pH by excreting the excess H+ into the urine. This H+ excretion is accompanied by Na+, K+, phosphate and water loss. This adds to the already severe water loss due to osmotic diuresis driven by the high glucose concentration in the blood (hyperosmolarity) and results in "diabetic coma" and a potentially fatal reduction in blood volume.
Tests for ketone bodies
Measurement of ketone bodies is useful in distinguishing between different types of DKA which require different treatment. Acetoacetate, usually the major ketone body, is easily detected by the maroon colour produced when it reacts with sodium nitroprusside at alkaline pH. This chemical is dried onto the paper pad at the end of KETOSTIX™ used for urine testing. However, about 10-15% of patients with DKA appear negative or only mildly positive when tested with KETOSTIX™. This is because some individuals convert nearly all their acetoacetate to b-hydroxybutyrate, which does not react with nitroprusside. If this is suspected, an alternative assay (immuno- or enzyme) for b-hydroxybutyrate must be carried out to confirm the diagnosis. Another group (typically older patients) presenting with coma and acidosis may also test negative for ketone bodies. They are described as hyperosmolar non-ketotic (HONK) as they have high glucose but no ketone bodies. In this case, acidosis is due mainly to lactic acid. All DKA cases require rehydration, but with HONK this must be done slowly. Hence, correct diagnosis is important.
Test for proteinuria
The incidence of renal complications increases with the duration of diabetes. Normally, urine contains no protein but as renal function deteriorates, protein can be detected. Since albumin is the predominant serum protein, detection of this protein indicates general proteinuria..
At pH values below its isoelectric point (the pH at which its overall charge due to surface ionisable amino acids is zero – about 5), albumin is positively charged and so it will bind anions (negatively charged ions). Indicator dyes, such as tetrabromophenol blue (TBB), change colour when they ionise.
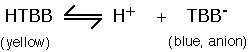
Both coloured forms are in equilibrium in solution, with the yellow form predominating at a pH just below the pK for the dissociation (3.5 in this case). However, if albumin is present, it binds the small amount of the blue anion and removes it from free solution, thus shifting the equilibrium to the right. More anion is produced and so on. The degree of colour shift from yellow (through green) to blue depends on the amount of protein bound blue-anion generated i.e. on the amount of albumin. This is the basis of the ALBUSTIX™ test for proteinuria.
Test for glycated haemoglobin
Glucose and some other sugars have the ability to react chemically with some of the amino acid side chains in proteins to form glycated proteins. Small amounts of these are present normally in blood. However, a persistent elevated blood glucose will lead to the accumulation of higher levels of glycated proteins such as albumin and haemoglobin. A particular modified haemoglobin called haemoglobin A1c (HbA1c) is quite stable with a half-life of 60-90 days. Therefore, the level of HbA1c is a good indicator of how well a patient has been managing his or her diabetes over a period of several weeks. HbA1c is normally 4-6% of total Hb. A high level (>7%) indicates persistent hyperglycaemia over that period and, therefore, poor management. An errant patient attending the diabetic clinic may try to fool the doctor by taking a dose of insulin just before the consultation to lower the blood glucose and by falsifying the glucose control diary. However, high HbA1c will identify the fraud. Therefore, a rapid assay that can be performed in a few minutes in the clinic while the patient is present is extremely valuable. A simple dipstick test that would differentiate between normal haemoglobin and HbA1c is not available. However, HbA1c can be rapidly separated from normal haemoglobin and measured by high performance liquid chromatography (HPLC).
Liquid chromatography
Liquid chromatography encompasses a variety of techniques that utilise as their basis the relative partition of solutes between a stationary phase and a mobile phase that passes through or across the stationary phase (Fig. 2). A solute that has a high affinity for the stationary phase (red sphere) will be retarded relative to a solute that remains in the mobile phase [i.e. has a low affinity for the stationary phase - (green sphere)]. Thus, different solutes may be separated on the basis of their partition between the two phases of the chromatographic system.

Figure 2: The basis of liquid partition chromatography
The different forms of liquid chromatography differ in the nature of the stationary and mobile phases. In the most common forms of chromatography, the stationary phase consists of an insoluble material (usually in beaded or granular form, e.g. cellulose or polystyrene) that is packed into a column and through which the mobile phase (containing the solutes) can flow.
High performance liquid chromatography
The degree of resolution achievable in liquid chromatography is in part dependent on the surface area of the stationary phase to which the mobile solutes are exposed. An increase in surface area can be achieved by decreasing the size of the particles comprising the stationary phase. This is turn leads to closer and tighter packing of the particles, so high pressure is required to pump the sample and the mobile phase through the column.. The tubing and columns used in HPLC (high performance or high pressure) liquid chromatography are usually made of stainless steel to withstand the pressure produced by the pumps. In the case of proteins, the highly resolved peaks from a complex mixture are normally visualised in the eluate coming out of the column by passing this though a continuous flow UV detector that measures the absorbance of the proteins in the ultraviolet range (due to the UV-absorbing amino acids tyrosine and tryptophan). Coloured proteins (e.g. haemoglobin) may also be detected by their absorbance in the visible region of the spectrum.
The system to be demonstrated employs combined reversed-phase and cation exchange chromatography followed by bichromatic spectrophotometric measurement (i.e. measurement of absorbance at a primary wavelength of 415 nm and a secondary wavelength 500 nm).
The urine samples
On the basis of the tests, you will determine which urine sample (A, B, C, D or E) corresponds to which case.